What You Ought to Know:
– The U.S. Meals and Drug Administration (FDA) has not too long ago issued a number of key clearances, marking important developments throughout diagnostics, surgical procedure, and distant affected person monitoring.
– These FDA clearances mirror a deepening integration of Synthetic Intelligence (AI) and superior expertise into medical workflows, promising better precision, lowered invasiveness, and expanded entry to care.
Diagnostics and Distant Monitoring: AI and Wearables at Scale
A serious pattern in current clearances focuses on leveraging AI and comfy wearables to reinforce diagnostic velocity and continuity of care.
Wearable Cardiopulmonary Monitoring
Hexoskin Receives 510(ok) Clearance for Lengthy-Time period ECG and Respiratory Monitoring
Carré Applied sciences Inc. (dba Hexoskin) obtained 510(ok) clearance for its Hexoskin Medical System (HMS) for steady long-term ECG, respiratory monitoring, and exercise in ambulatory sufferers. This method, which features a good biometric shirt, turns into one of many first medical-grade wearable programs able to long-term ECG and respiratory measurements outdoors the clinic, remodeling distant care and decentralized medical trials. The expertise allows physicians to evaluate arrhythmias (like atrial fibrillation) and respiration price patterns with steady knowledge assortment.
For medical analysis, HMS represents a significant step ahead. With FDA clearance, the system can now assist decentralized trials, permitting investigators to seize high-resolution, real-world physiological knowledge and develop AI-driven digital biomarkers throughout cardiology, pulmonology, neurology, and uncommon ailments.
AI-Pushed Cardiac and Aortic Imaging
FDA clearances solidify AI’s function in cardiovascular danger administration:
RapidAI Receives FDA Clearance for Fast Aortic, Bringing Deep Medical AI to Aortic Illness Administration
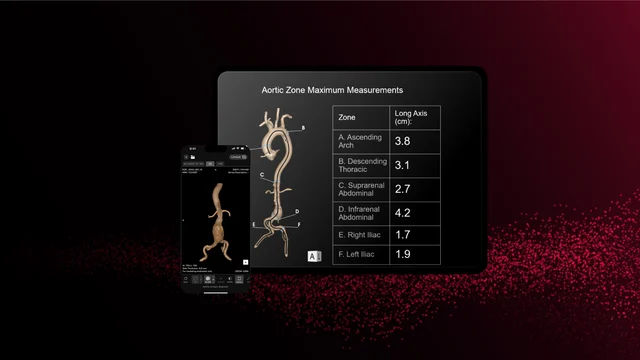
RapidAI earned FDA clearance for Aortic Administration, a part of its Fast Aortic product. This deep medical AI answer transforms the acute evaluation and longitudinal administration of aortic illness. It mechanically generates important measurements (together with zonal maximums and landmark metrics), produces 3D reconstructions, and tracks anatomical modifications over time to help in figuring out and monitoring pathology from the aortic arch to the iliacs.
In contrast to conventional AI triage instruments, Fast Aortic is engineered to assist end-to-end affected person administration: screening, prognosis, remedy planning, and surveillance. The system’s capacity to course of all CT scans containing the aorta—whether or not distinction or non-contrast—expands its utility throughout emergency, inpatient, and outpatient settings.
Clinicians stand to profit from lowered cognitive burden, quicker learn occasions, and improved accuracy. Surgeons can leverage exact visualizations for pre-procedural planning, whereas well being programs acquire a unified workflow built-in via the Fast Edge Cloud and Fast Navigator Professional.
Circle CVI Receives 510(ok) Clearance for AI-Enabled Coronary Plaque Evaluation
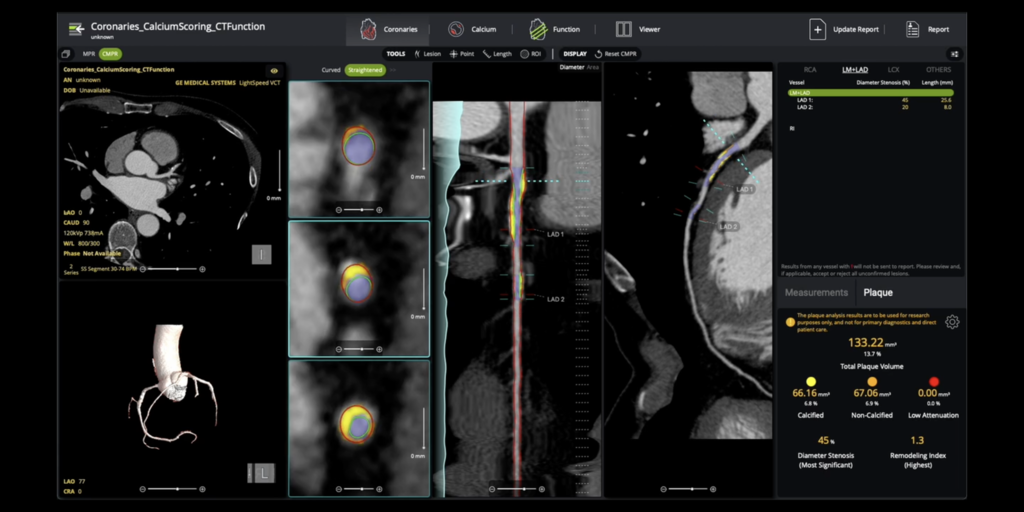
Circle Cardiovascular Imaging Inc. (Circle CVI) obtained 510(ok) clearance for its cvi42 | Plaque answer for complete coronary plaque evaluation. This AI-enabled, on-premise expertise quantifies whole, calcified, and non-calcified plaque, supporting exact danger stratification. The clearance coincides with a brand new Class I CPT code (75XX6) taking impact in January 2026, solidifying plaque quantification as normal medical care.
Bunkerhill Well being Secures FDA Clearance for First AI to Detect Mitral Annular Calcification on Routine Chest CT

Bunkerhill Well being achieved the first-ever FDA clearance for an AI algorithm—Bunkerhill MAC—to detect and quantify mitral annular calcification (MAC) on routine, non-gated chest CT scans. MAC is an often-overlooked discovering linked to elevated cardiovascular danger and procedural problems.
Built-in into Bunkerhill’s Carebricks platform, the instrument leverages FDA-cleared AI and large-language-model-based reasoning to assist follow-up choices inside cardiology, major care, and structural coronary heart packages. The clearance displays the FDA’s broader confidence in AI that elevates incidental findings into actionable medical insights.
Neuroscience and Ache Administration
A number of clearances goal mind perform and continual ache:
QuantalX Secures De Novo Clearance for Delphi-MD, a First-of-Its-Variety Purposeful Neuro-Imaging Expertise
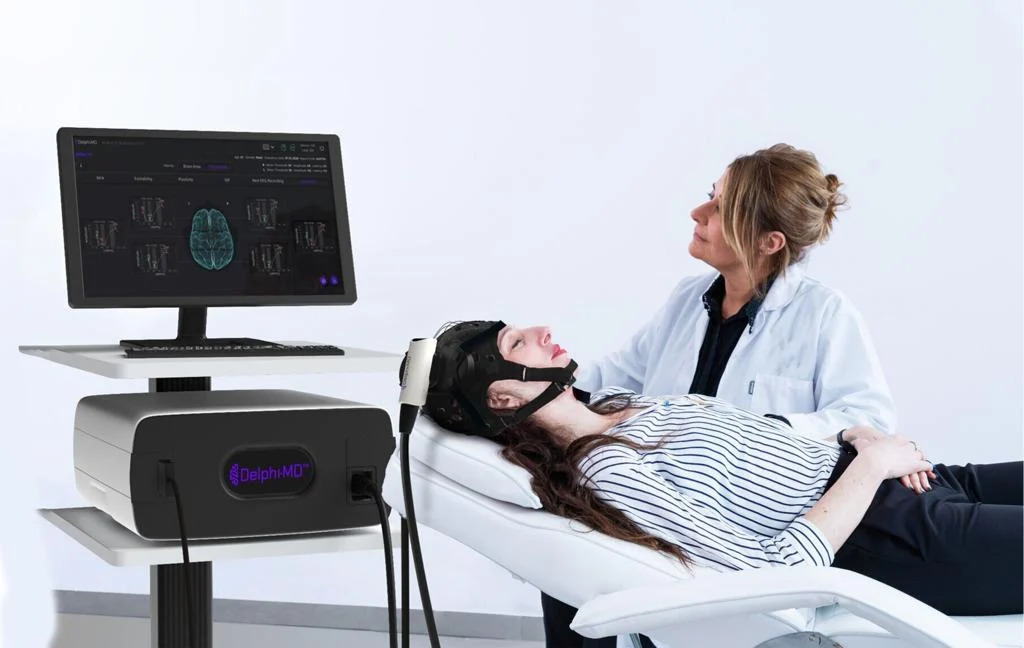
QuantalX Neuroscience was granted De Novo classification for its Delphi-MD™ System, establishing a brand new modality of practical neuro-imaging (FNI). Delphi-MD combines non-invasive Transcranial Magnetic Stimulation (TMS) with electroencephalography (EEG) to benchmark mind community perform in opposition to a normative database.
Delphi-MD offers physicians a benchmarked evaluation of mind perform utilizing an FDA-cleared normative database of wholesome adults. This creates a novel medical modality able to monitoring cognitive decline, evaluating neurological interventions, and aiding illness administration throughout neurodegenerative, traumatic, or pain-related situations.
Magstim Achieves FDA Clearance for Non-Invasive Magnetic Stimulation for Continual Ache
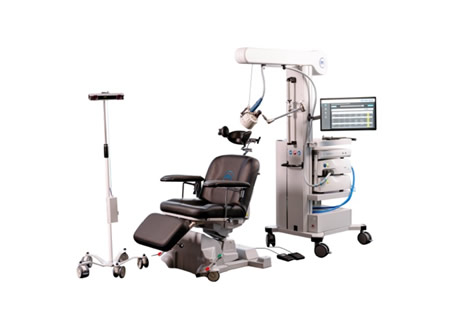
The FDA cleared Magstim Magnetic Stimulation for the remedy of continual ache, offering a clinically confirmed, non-invasive, and drug-free possibility. The expertise modulates peripheral nerve pathways utilizing magnetic pulses, reaching deeper nerves with out invasive implants or prescribed drugs.
Magstim’s expertise—cited in additional than 15,000 scientific research—supplies deeper nerve stimulation in contrast with conventional TENS or surface-level gadgets, providing an necessary possibility for sufferers for whom typical therapies are insufficient.
Surgical Robotics and Precision Orthopedics
Improvements in robotic help intention to enhance precision and cut back trauma:
Zimmer Biomet Secures 510(ok) Clearance for ROSA Knee With OptimiZe, Increasing Robotic Precision in Orthopedics
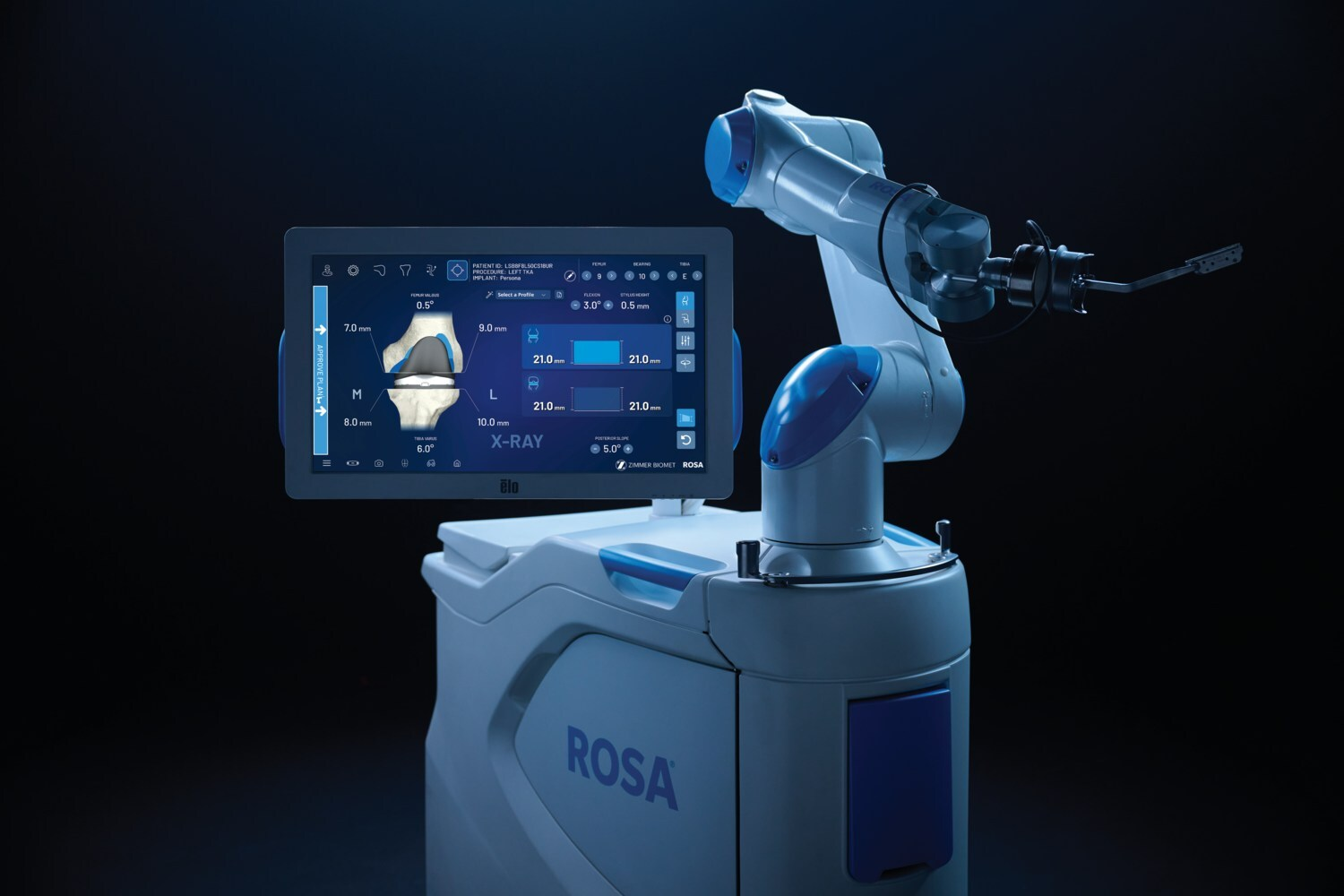
Zimmer Biomet gained 510(ok) clearance for ROSA® Knee with OptimiZe™, an enhanced model of its robotic system for whole knee alternative surgical procedure. The expertise provides custom-made clever surgical planning and options like OptimiZe Kinematic Alignment™—the trade’s solely automated kinematic alignment characteristic—to make sure correct, reproducible outcomes based mostly on affected person anatomy and surgeon preferences.
Built-in with ZBEdge® Analytics, ROSA Knee with OptimiZe allows data-driven choices, real-time intraoperative insights, and steady efficiency analysis. A focused launch will start later this 12 months, with U.S. industrial availability anticipated in early 2026.
Levita Magnetics Positive factors Pediatric Clearance for Magnetic Surgical System (MSS)
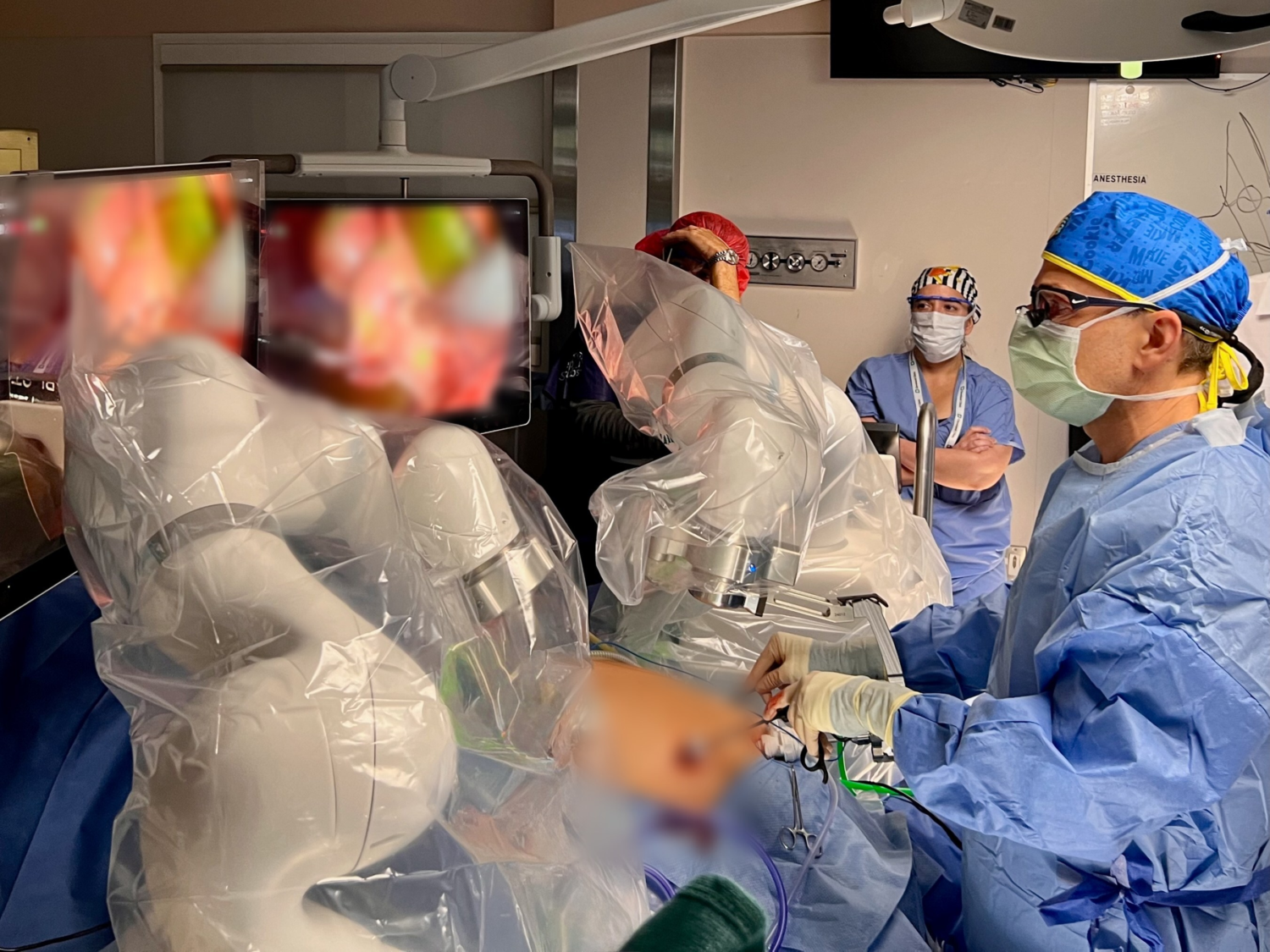
Levita® Magnetics achieved FDA clearance for its Magnetic Surgical System (MSS) for pediatric sufferers. This expertise makes use of exterior magnets to regulate inside surgical devices, lowering the variety of incisions wanted for procedures like laparoscopic cholecystectomy, which is important for minimizing trauma and scarring in youngsters.
Cleveland Clinic Youngsters’s turned the primary middle to carry out a pediatric case utilizing this expertise. For youthful sufferers, minimizing tissue trauma is important: fewer ports can result in quicker restoration, decreased ache, lowered scarring, and decrease complication dangers.
FDA Approves First-Ever Robotic Surgical Research for Alzheimer’s Illness Intervention

MMI (Medical Microinstruments, Inc.) obtained FDA Investigational System Exemption (IDE) approval for a medical examine—REMIND—utilizing the Symani® Surgical System for a novel microsurgical intervention for Alzheimer’s illness. The examine goals to reestablish lymphatic drainage pathways within the deep cervical lymph nodes to doubtlessly enhance the clearance of dangerous proteins. The process requires supermicrosurgical precision, working on vessels as small as 0.2mm.
Wound Care and Diabetes Administration
Fast Nexus Earns 510(ok) Clearance for Hemastyl, a Breakthrough in Continual Wound Remedy
Fast Nexus Nanotech Wound Options, Inc. obtained FDA 510(ok) clearance for its Hemastyl gel machine. The gel is the primary machine to instantly deal with the periwound tissue atmosphere—the residing edge chargeable for stalled restoration—to revive situations for wound closure and assist sufferers keep away from amputations.
Fast Nexus plans to pursue FDA Breakthrough System Designation, which might expedite protection and reimbursement pathways, making this remedy obtainable to hundreds of thousands of high-risk sufferers nationwide. The clearance is a landmark second for wound therapeutic—a subject lengthy dominated by symptomatic slightly than causative remedy approaches.
Tandem Diabetes Care Positive factors FDA Clearance for Tandem Mobi App for Android Customers
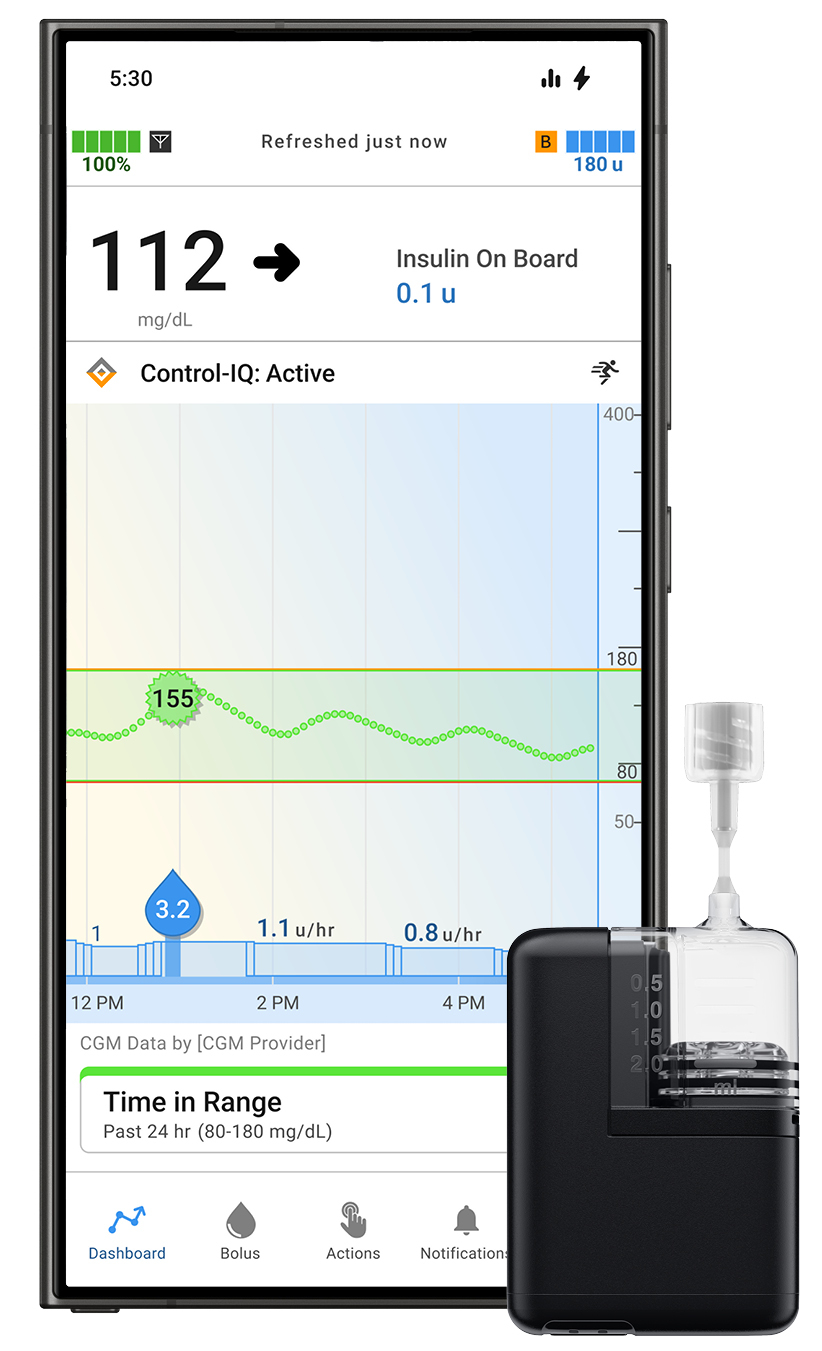
Tandem Diabetes Care, Inc. obtained FDA clearance for the Android model of its Tandem Mobi cellular app. This enables Android customers to handle their diabetes instantly from their appropriate smartphone utilizing the Tandem Mobi automated insulin supply system, which is powered by Management-IQ+ expertise.
The clearance considerably expands entry for sufferers preferring Android gadgets, enhancing usability and affected person adherence. A restricted launch is anticipated in late 2025, with broad industrial availability in early 2026.






